Title Page
-
Site conducted
-
Conducted on
-
Prepared by
-
Location
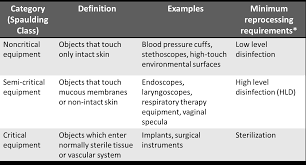
General
-
Observe delivery of a soiled device. Is the device in an impermeable container or bag, labeled with a biohazard sticker?
-
Observe the use of personal protective equipment (PPE) when staff performs high-level disinfection. Is the use of PPE in accordance with the manufacturer's IFU and policy?
-
Is the location and use of the high-level disinfecting system and chemicals safe for patients and staff? (Examples: Are the surfaces stable to prevent spillage or tipping; are chemicals located at a height to promote worker safety; is cleaning and disinfection performed outside the immediate proximity to the patient/treatment area; are chemicals located in a secured area and not accessible to patients or visitors?)
-
Are containers for high-level disinfectants properly labeled when removed from the original container? (Note: Labels should identify the contents and hazard warnings. Review appropriate policy for additional labeling requirements.)
-
Observe all unopened containers of disinfectant. Is the expiration date(s) and are the date(s) within range?
-
Ask staff to locate the Safety Data Sheet for the chemical disinfectant in use. Can staff locate this information without difficulty or delay?
-
Ask staff to describe the process to follow in the event of a spill. Can staff describe the process to follow immediately post-spill, including whom to notify and how? (If the spill is less than the size of a dinner plate, use the spill kit and proper PPE. If the spill is larger than a dinner plate, secure the area and call EVS).
-
Observe the location where the equipment or devices are stored following high-level disinfection. Is the equipment or device stored in a manner to prevent contamination? (Examples: Individual devices should be stored in a clean area and preferrably covered with a protective sleeve/wrap. Endoscopes should be hung vertically in a clean, ventilated cabinet with tips not touching the bottom of the cabinet.)
-
If cleaned devices are stored in this department, evaluate the storage area/cabinet. Are the devices stored according to manufacturer's recommendations? (Ex. Scopes should be stored in a manner that enables them to hang freely, in a cabinet that is monitored for temperature and humidity).
-
If the location has clean scope storage, is the temperature and humidity documented for each day?
-
Ask staff to describe the process for managing a device that has not been used for an extended period of time after disinfection. Score compliant if the staff state that the device is reprocessed after 7 days (?) if not used.
-
Review staff education files of at least 1 individual who performs high-level disinfection. Does documentation include education and competence assessment of the high-level disinfection process being performed, including specifics for the item being disinfected and the disinfectant being used? Was competence assessment documented during orientation and at defined frequencies?
-
Ask staff to locate the nearest eyewash station. Can staff reach the eyewash within 10 seconds/55 feet, with an unobstructed path?
-
Review documentation of eyewash checks on the tag at the station for the last month. Does documentation include weekly checks in accordance with policy?
Trophon
-
Wipe the ultrasound probe prior with a disinfectant wipe and then with a low-linting dry wipe (plug to probe)
-
When placing probe into Trophon unit, make sure the tip of the probe is above the line, the probe is not touching the sides of the unit, and the cord of the probe is coiled at the top of the unit
-
Confirm that the unit is working properly with the chemical indicator
-
Check the log has been filled out
GUS
-
Ask staff to describe the minimal soak/cycle time for the disinfectant used and the device being disinfected. Review disinfection documentation. Does the staff state the appropriate soak/cycle time and document this correctly?
-
Ask staff to describe the process to rinse the device after disinfection. Review the policy and manufacturer's instructions for use. Does the staff's description match the manufacturer's instructions for use for the process?
-
Check all containers of disinfectant efficacy test strips for the expiration date located on the container. Are all expiration dates within an acceptable range?
-
Observe all open containers of test strips. Are all containers labeled with the new expiration date, once opened, in accordance with manufacturer's guidelines?
-
Determine if the test strip manufacturer requires a quality control process when initially opening a container of test strips. If quality control is required, does documentation support this process was followed for all open containers of test strips?
-
Discuss with staff the process to check the disinfectant for efficiency with test strips. Does the process, as described - and a review of documentation - indicate that the disinfectant is checked for efficacy with each use, in accordance with policy and manufacturer's instructions for use?
-
Determine if the disinfectant manufacturer has defined a minimal effective temperature or recommended temperature range for the disinfectant. If so, does documentation indicate a process to ensure the disinfectant temperature is appropriate?
-